How Long Will 10mg of Retatrutide Last? Dosage, Duration, Research Guide 2026

In the rapidly evolving world of metabolic peptide research, few compounds have generated as much scientific attention as retatrutide 10mg. Researchers, biochemists, and institutions across the United States, Europe, and Asia-Pacific are actively studying this investigational triple-agonist peptide and for good reason. Its unique mechanism of simultaneously targeting three distinct metabolic receptors makes it one of the most complex and promising compounds in current Phase 3 clinical research.
But one question dominates the research community more than almost any other: how long will 10mg of retatrutide last? Whether the question refers to the compound’s half-life, the duration of receptor activity, or how long a 10mg vial supply lasts for a research protocol, the answer requires unpacking several layers of pharmacokinetics, dosing schedules, and research design.
This guide written from a research-oriented perspective and aligned with the latest available data as of early 2026 provides a comprehensive, evidence-informed breakdown of retatrutide 10mg, its mechanism, expected duration parameters, and everything researchers need to understand before working with this compound.
What Is Retatrutide? Understanding the Triple Agonist Compound
Retatrutide (also referenced by its research code LY3437943) is an investigational peptide designed as a triple receptor agonist, meaning it activates three distinct hormonal pathways simultaneously:
| Receptor Targeted | Primary Function | Role in Metabolic Research |
|---|---|---|
| GLP-1 (Glucagon-Like Peptide-1) | Stimulates insulin secretion and suppresses appetite | Reduces caloric intake and improves glucose control |
| GIP (Glucose-Dependent Insulinotropic Polypeptide) | Enhances insulin response and supports fat metabolism | Works synergistically with GLP-1 to amplify metabolic effects |
| Glucagon Receptor | Increases energy expenditure and promotes lipolysis | Supports fat oxidation and boosts thermogenesis |
This triple mechanism is what separates retatrutide from single-agonist compounds like semaglutide and even dual-agonists like tirzepatide. In clinical research, subjects receiving retatrutide demonstrated significantly greater reductions in body weight compared to those receiving other GLP-1-class compounds making it one of the most studied peptides in this category.
For researchers interested in how triple-agonist peptides compare to other GLP-class compounds, our detailed breakdown of how long semaglutide side effects last provides important comparative context.
Retatrutide 10mg: Key Research Facts at a Glance
Before diving deep into duration and dosage specifics, here is a high-level fact table covering the core attributes of retatrutide 10mg as understood from current clinical research:
| Research Parameter | Detail |
|---|---|
| Compound Class | Triple GLP-1/GIP/Glucagon Receptor Agonist |
| Research Status (2026) | Phase 3 Clinical Trials (Not FDA Approved) |
| Administration Route | Subcutaneous injection (research protocols) |
| Reported Half-Life | Approximately 6–7 days |
| Research Dosing Frequency | Once-weekly (in clinical trial settings) |
| Common Research Vial Size | 10 mg lyophilized powder |
| Storage Recommendation | Lyophilized: 2–8°C; Reconstituted: −20°C for long-term |
| Availability Status | Research-grade; not approved for human use |
| Primary Research Area | Obesity, metabolic syndrome, type 2 diabetes model research |
| Manufacturer Research Code | LY3437943 (Eli Lilly investigational compound) |
How Long Will 10mg of Retatrutide Last? Breaking Down the Duration Question
This is the core question this guide addresses and the answer depends entirely on what aspect of “lasting” you’re asking about. There are three distinct ways researchers interpret this question:
1. How Long Does Retatrutide Stay Active in a Research Model? (Pharmacokinetic Duration)
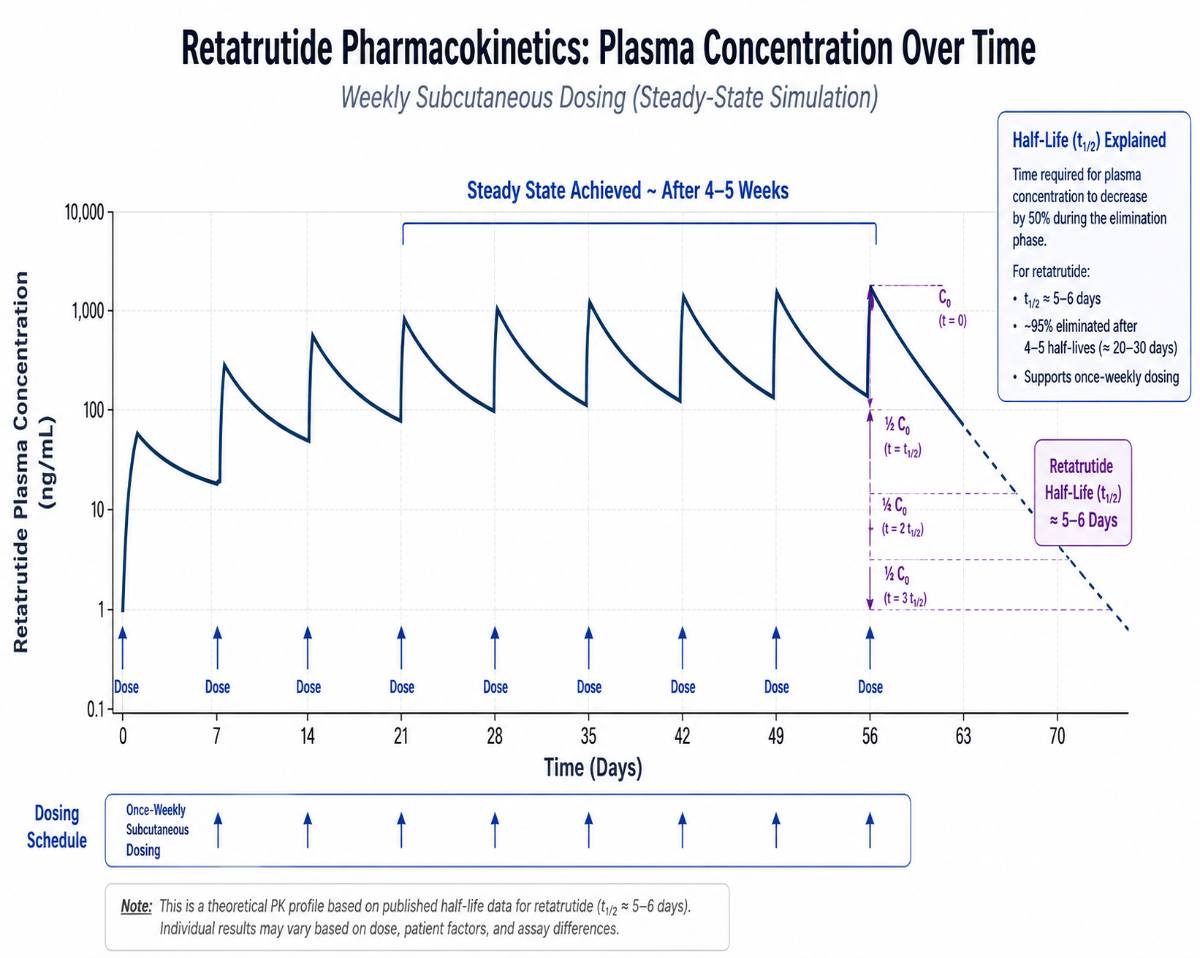
Based on available Phase 2 and Phase 3 clinical trial data, retatrutide demonstrates a plasma half-life of approximately 6 to 7 days. This extended half-life is achieved through a fatty acid chain modification (similar to semaglutide’s structural design) that allows the compound to bind to albumin in the bloodstream, slowing its clearance.
In practical pharmacokinetic terms:
- Half-life: ~6–7 days
- Time to reach steady-state plasma concentration: approximately 4–6 weeks with weekly dosing
- Duration of detectable receptor activity: up to 10–14 days post-administration in some models
- Full elimination (5 half-lives): approximately 30–42 days
This means that, in a research protocol administering retatrutide once weekly, the compound does not fully clear between doses, by design, as accumulation toward steady-state is part of the intended pharmacological profile.
2. How Long Does a 10mg Vial of Retatrutide Last in a Research Protocol?
This is the supply-side interpretation of the question and the answer depends on the research dosing schedule.
In Phase 2 clinical trials, researchers used escalating dose protocols. Below is a representation of how a 10mg research vial might be allocated based on trial-modeled dosing schedules:
| Weekly Research Dose | Number of Doses from 10 mg Vial | Approximate Supply Duration |
|---|---|---|
| 0.5 mg/week (initiation phase) | 20 doses | ~20 weeks |
| 1.0 mg/week | 10 doses | ~10 weeks |
| 2.0 mg/week | 5 doses | ~5 weeks |
| 4.0 mg/week | 2–3 doses | ~2–3 weeks |
| 8.0 mg/week | 1 dose | ~1 week |
Clinical trials typically began at 0.5–1.0 mg weekly, escalating gradually over 4–8 weeks before reaching higher research doses. This titration is designed to minimize adverse events during the study period.
For researchers comparing this protocol design to other peptide compounds, the CJC-1295 Ipamorelin Research Guide at Ageless Vitality Peptides demonstrates how similar escalation principles are applied across other peptide categories.
3. How Long Do the Research Effects of Retatrutide Last After Cessation?
Based on Phase 2 trial data (published in the New England Journal of Medicine, 2023), subjects who discontinued retatrutide experienced gradual weight regain over several months consistent with the behavior observed with other GLP-1 class compounds. The receptor-level effects are not permanent, as the compound does not alter underlying genetic expression.
Key observations from cessation research:
- Receptor activity reduces proportionally with plasma clearance (~6–7 day half-life)
- Metabolic effects (appetite suppression, insulin sensitization) diminish over 2–4 weeks post-last dose
- Full pharmacological washout is estimated at approximately 4–6 weeks after the final administration
Retatrutide 10mg Mechanism: The Triple Agonist Advantage Explained
Understanding why retatrutide 10mg produces such pronounced effects in research requires a deeper look at each receptor pathway it engages.
GLP-1 Receptor Agonism
GLP-1 receptor activation underlies the compound’s appetite-suppressing properties. When GLP-1 receptors are activated:
- The hypothalamus receives satiety signals, reducing hunger drive
- Gastric emptying is slowed, prolonging post-meal fullness
- Pancreatic beta cells increase insulin secretion in a glucose-dependent manner
This is the same pathway targeted by semaglutide and liraglutide. In comparison research, understanding the differences between tesamorelin vs sermorelin also illustrates how receptor-specific targeting produces different metabolic outcomes.
GIP Receptor Agonism
GIP agonism amplifies the GLP-1 effect in several key ways:
- Enhances insulin secretion synergistically
- Reduces the nausea associated with GLP-1 agonism alone (a significant finding)
- May directly promote adipose tissue lipolysis
The inclusion of GIP agonism is one of the reasons retatrutide’s side effect profile in trials was notably better tolerated at equivalent or higher weight-reduction efficacy compared to GLP-1-only compounds.
Glucagon Receptor Agonism
This third pathway is what truly differentiates retatrutide from all other approved or near-approved compounds. Glucagon receptor activation:
- Increases hepatic glucose output (managed by the concurrent insulin-sensitizing effects)
- Promotes thermogenesis and caloric expenditure at rest
- Accelerates fatty acid oxidation in the liver
In research models, the net result has been a greater caloric deficit effect than with GLP-1 or dual-agonists, contributing to the significant weight-reduction percentages observed in Phase 2 trials.
Retatrutide 10mg vs. Other Peptides: Comparative Research Overview
Researchers frequently compare retatrutide to other peptides in the metabolic and recovery research space. Here is how it stacks up against some of the most studied compounds:
| Compound | Receptor Target | Primary Research Area | Half-Life |
|---|---|---|---|
| Retatrutide 10mg | GLP-1 + GIP + Glucagon | Metabolic, obesity research | ~6–7 days |
| Semaglutide | GLP-1 only | Metabolic, glucose research | ~7 days |
| Tirzepatide | GLP-1 + GIP | Metabolic, diabetes research | ~5 days |
| BPC-157 5mg | Receptor-independent (pleiotropic) | Tissue healing, GI research | Hours |
| TB-500 (Thymosin Beta-4) | Actin-sequestering, angiogenic pathways | Wound repair, recovery research | Days |
| CJC-1295 / Ipamorelin Blend | GHRH + Ghrelin receptor | Growth hormone axis research | Hours–Days |
| IGF-1 LR3 | IGF-1 receptor | Anabolic, cellular proliferation | ~20–30 hours |
While retatrutide occupies a unique position in metabolic research, many researchers also work with peptides targeting different systems. For those studying tissue repair protocols alongside metabolic research, compounds like BPC-157 5mg and TB-500 Thymosin Beta-4 from Ageless Vitality Peptides represent complementary research avenues in connective tissue and recovery science.
Retatrutide 10mg: Reconstitution and Storage Research Notes
For laboratory researchers working with retatrutide 10mg in lyophilized (freeze-dried powder) form, proper handling is essential for data integrity.
Reconstitution Guidelines (Research Settings)
The standard research reconstitution protocol for lyophilized peptides involves bacteriostatic water. For a 10mg vial:
| Target Concentration | Bacteriostatic Water Volume | Resulting Concentration |
|---|---|---|
| 1 mg/mL | 10 mL | 1,000 mcg/mL |
| 2 mg/mL | 5 mL | 2,000 mcg/mL |
| 5 mg/mL | 2 mL | 5,000 mcg/mL |
These concentrations are relevant for precise research dosing calculations and should be documented in research protocols.
Storage Duration After Reconstitution
| Storage Condition | Expected Stability |
|---|---|
| Lyophilized, 2–8°C (refrigerated) | 12–24 months (manufacturer-dependent) |
| Lyophilized, −20°C (frozen) | 24+ months |
| Reconstituted, 2–8°C | 28–30 days |
| Reconstituted, −20°C | 3–6 months (some degradation expected) |
Proper storage directly impacts the question of “how long will 10mg of retatrutide last” from a stability standpoint improper storage can significantly reduce peptide integrity and research validity.
Clinical Trial Data Highlights: What Phase 2 Research Revealed
While Phase 3 data continues to accumulate as of 2026, the Phase 2 results (published in 2023) provided compelling findings that drove continued development:
Body Weight Reduction in Phase 2 Subjects
| Retatrutide Dose (Weekly) | Mean Body Weight Reduction (48 weeks) |
|---|---|
| 1 mg | ~8.7% |
| 4 mg | ~17.1% |
| 8 mg | ~22.8% |
| 12 mg | ~24.2% |
These figures represent some of the highest weight reduction percentages observed in any pharmacological obesity intervention to date, eclipsing the approximately 15% seen with semaglutide 2.4mg in STEP trials.
Cardiometabolic Markers (Phase 2)
- Systolic blood pressure: reduced by 4–8 mmHg across dose groups
- Fasting triglycerides: reduced by up to 30% in higher dose cohorts
- HbA1c: significant reductions in subjects with elevated baseline glucose
- Waist circumference: significant reductions correlated with visceral fat loss
Common Side Effects Observed in Retatrutide Research
Consistent with other GLP-1 class compounds, the most commonly observed adverse events in retatrutide clinical research include:
| Side Effect | Frequency Reported | Notes |
|---|---|---|
| Nausea | Most common | Dose-dependent; typically resolves with titration |
| Vomiting | Common | Associated with higher doses; generally transient |
| Diarrhea | Common | GI tract adaptation; often self-limiting |
| Constipation | Moderate frequency | Possibly more common than with GLP-1 alone |
| Decreased appetite | Intended effect; also reported as adverse event by some | Expected pharmacodynamic response |
| Eructation (belching) | Less common | Associated with GIP agonism |
| Injection site reactions | Low frequency | Standard subcutaneous injection reactions |
The addition of GIP agonism in retatrutide appears to reduce the severity of nausea compared to GLP-1-only compounds at equivalent efficacy a finding that distinguishes it from semaglutide. This is consistent with the observation that tirzepatide (a GLP-1/GIP dual agonist) also causes less nausea than semaglutide at comparable efficacy doses.
For researchers exploring related peptide side effect profiles, our resource on how long semaglutide side effects last provides a useful GLP-1 class reference framework.
Related Peptide Research at Ageless Vitality Peptides
Researchers exploring retatrutide 10mg often work within broader metabolic and tissue repair research programs. Ageless Vitality Peptides provides a range of research-grade peptides to support diverse research objectives:
For Growth Hormone Axis Research
The CJC-1295 5mg / Ipamorelin 5mg Blend (10mg) is one of the most widely used growth hormone secretagogue combinations in research. Its synergistic pairing of a GHRH analog (CJC-1295) with a ghrelin mimetic (Ipamorelin) produces sustained GH pulse amplification distinct from but complementary to metabolic research involving retatrutide.
For Tissue Repair and Recovery Research
- BPC-157 5mg — A Body Protection Compound fragment studied extensively for its pleiotropic healing effects on GI tissue, tendons, and neurological repair.
- TB-500 Thymosin Beta-4 Fragment 5mg — A synthetic analog of Thymosin Beta-4, studied for angiogenesis, tissue remodeling, and accelerated repair.
- BPC-157 5mg + TB-500 5mg Blend (10mg) — A popular combined research formulation studied for synergistic tissue healing; researchers have explored the BPC-157 and TB-500 combination for complementary mechanisms.
For IGF and Anabolic Research
The IGF-1 LR3 before-and-after research data documented by Ageless Vitality Peptides provides context for researchers studying downstream effects of GH-axis stimulation — often relevant in programs that also investigate metabolic compounds like retatrutide.
How to Source Retatrutide 10mg for Research: What to Look For
Given that retatrutide is an investigational compound not approved by the FDA, researchers must be highly selective about their supplier. Key quality indicators include:
Supplier Verification Checklist
| Quality Indicator | What to Look For |
|---|---|
| Certificate of Analysis (COA) | Independent third-party lab testing, HPLC purity data |
| Mass Spectrometry Verification | Confirms molecular weight matches retatrutide |
| Lyophilized Form | Indicates proper manufacturing and preservation |
| Research-Only Labeling | Confirms not represented for human use |
| Transparent Company Information | Physical address, contact information, regulatory disclosure |
| Secure Ordering Platform | SSL encryption, privacy-compliant checkout |
Ageless Vitality Peptides provides retatrutide 10mg for research procurement with documentation aligned with laboratory research standards. All products are provided as chemical reagents for research purposes only.
Retatrutide Research in 2026: What to Expect Next
As Phase 3 clinical trials continue their enrollment and data collection, the research community is monitoring several key milestones:
Anticipated 2026–2027 Developments:
- Phase 3 primary endpoint data readouts expected from multiple trial cohorts
- Potential NDA (New Drug Application) submission to the FDA pending positive Phase 3 outcomes
- Comparative effectiveness research against tirzepatide (Mounjaro/Zepbound) is anticipated in peer-reviewed literature.
- Cardiovascular outcomes trial (CVOT) data expected to emerge in this window.
- Potential expansion research into non-alcoholic fatty liver disease (NAFLD/NASH)
The compound’s trajectory suggests that retatrutide may become a significant point of reference in metabolic research for years to come — making current research into its mechanisms and pharmacokinetics particularly valuable for the scientific community.
[Add Image Here: Timeline infographic — Retatrutide clinical development milestones from Phase 1 (2021) through Phase 3 (2024–2026) and projected NDA submission window]
Key Takeaways on How Long Will 10mg of Retatrutide Last
To consolidate the core answer this guide has built toward:
| Duration Question | Answer |
|---|---|
| Plasma half-life of retatrutide | ~6–7 days |
| Time to steady-state (weekly dosing) | 4–6 weeks |
| Full pharmacological washout | ~30–42 days |
| How long 10 mg vial lasts at 0.5 mg/week | ~20 weeks |
| How long 10 mg vial lasts at 1 mg/week | ~10 weeks |
| How long 10 mg vial lasts at 2 mg/week | ~5 weeks |
| Reconstituted stability (refrigerated) | 28–30 days |
| Lyophilized stability (refrigerated) | 12–24 months |
The answer is not a single number it depends on which aspect of duration matters most to your research design: pharmacokinetics, supply management, or post-cessation washout timelines.
Frequently Asked Questions (FAQs)
How long will 10mg of retatrutide last in a research protocol?
The duration depends entirely on the dosing schedule used in the research design. At 0.5mg per week (the lowest initiation dose used in Phase 2 trials), a 10mg vial would provide approximately 20 weeks of supply. At 2mg per week, the same vial would last approximately 5 weeks. Researchers should calculate based on their specific protocol requirements.
What is the half-life of retatrutide?
Based on Phase 2 clinical trial pharmacokinetic data, retatrutide has an estimated plasma half-life of approximately 6–7 days. This extended half-life is enabled by fatty acid chain modification, allowing albumin binding and slower renal clearance.
How does retatrutide differ from semaglutide?
Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously. Semaglutide targets only the GLP-1 receptor. In clinical research, retatrutide has demonstrated greater percentage body weight reduction (up to 24.2% at 48 weeks) compared to semaglutide’s approximately 15% in STEP trial data though direct head-to-head trials are ongoing.
What are the most commonly reported side effects of retatrutide in research?
The most commonly reported adverse events in retatrutide clinical trials include nausea, vomiting, diarrhea, and constipation consistent with the GLP-1 agonist class. The GIP component appears to reduce nausea severity compared with GLP-1 monotherapy at equivalent efficacy, a distinguishing feature of the compound.
Can retatrutide 10mg be used with other peptides in research?
While combination research is outside the scope of approved protocols, researchers studying multiple peptide compounds in independent models should be aware of the distinct receptor systems involved. Retatrutide targets metabolic pathways (GLP-1/GIP/Glucagon), while compounds like BPC-157 and TB-500 operate through entirely different tissue repair mechanisms. Any combined research design should follow appropriate institutional guidelines.
How should reconstituted retatrutide be stored?
Reconstituted retatrutide should be stored at 2–8°C (standard refrigerator temperature) and used within 28–30 days for optimal integrity. For long-term storage of reconstituted compound, −20°C is recommended with the caveat that some degradation may occur over 3–6 months.
Is retatrutide available for purchase?
Research-grade retatrutide 10mg is available through specialized peptide research suppliers like Ageless Vitality Peptides as a chemical reagent for research purposes. It is not available as a prescription medication or supplement, and purchases must comply with applicable regulations.
What is the molecular weight of retatrutide?
Retatrutide has a molecular weight of approximately 4,810 Da (Daltons), consistent with its structure as a fatty acid-conjugated peptide analog.
Disclaimer: All content on this page is for informational purposes related to research only. Ageless Vitality Peptides products are chemical reagents not intended for human use. The FDA has not evaluated the statements made and they are not intended to diagnose, treat, cure, or prevent any disease. Always comply with local regulations when purchasing research compounds.