How to Reconstitute Peptides: Step-by-Step Guide for Researchers (2026)
If you’ve ever opened a peptide vial and wondered exactly what to do next, you’re not alone. Peptide reconstitution is one of the most important and most misunderstood steps in peptide research. Get it right, and your research compound maintains its full structural integrity and potency. Get it wrong, and you may damage the peptide’s delicate amino acid chain before the study even begins.
This guide is intended for researchers, lab professionals, and members of the scientific community who need a reliable, evidence-based reference on how to reconstitute peptides . Whether you’re working with growth hormone-releasing peptides, healing peptides, or metabolic research compounds, the fundamentals of reconstitution are the same.
Peptides are supplied as lyophilized (freeze-dried) powders. This preserves their stability during shipping and long-term storage. Before use in any research context, they must be reconstituted meaning they must be dissolved in a sterile liquid. The choice of solvent, the technique used, the concentration achieved, and how the solution is stored afterward all directly affect research validity and compound integrity.
According to peer-reviewed biochemistry literature, lyophilized peptides can lose up to 30% of their bioactivity if reconstituted incorrectly particularly if shaken rather than swirled, or if the wrong solvent is used. For researchers working with compounds like BPC-157, TB-500, or CJC-1295/Ipamorelin blends, this is especially relevant.
What Is Peptide Reconstitution?
Reconstitution is the process of adding a sterile liquid (solvent) to a lyophilized peptide powder to create a solution ready for research use. The goal is to dissolve the peptide completely without damaging its molecular structure.
Lyophilization the freeze-drying method used to preserve peptides removes all water from the compound under vacuum. This gives the powder excellent shelf life but means the peptide must be re-dissolved before it can be used in any biological research context.
The Word “reconstitute” literally means to restore to a former state. In peptide science, this means returning a dry, fragile amino acid chain to a stable aqueous solution while preserving every bond and residue that makes it biologically relevant to research.
| Term | Definition |
|---|---|
| Lyophilized | Freeze-dried; most peptides arrive in this powder state |
| Bacteriostatic Water | Sterile water with 0.9% benzyl alcohol; the most common reconstitution solvent |
| Sterile Water for Injection | Pure sterile water; no preservative; shorter shelf life after reconstitution |
| Acetic Acid Solution | Used for peptides that don’t dissolve well in water (e.g., some GHRPs) |
| Reconstitution | Dissolving lyophilized peptide into a liquid solvent |
| Concentration | Amount of peptide per volume of solvent (e.g., mg/mL) |
| Vial | The sealed glass container holding the peptide |
Choosing the Right Solvent for Reconstitution
The choice of solvent is critical. Using the wrong liquid can alter the peptide’s pH, cause aggregation, or destroy its structural integrity.
Bacteriostatic Water — The Gold Standard
Bacteriostatic water is sterile water containing 0.9% benzyl alcohol as a preservative. This preservative inhibits bacterial growth, making the reconstituted solution stable for up to 28–90 days when refrigerated, depending on the specific peptide.
Why is bacteriostatic water preferred?
- Inhibits microbial contamination between uses
- Compatible with most peptides
- Multi-dose vial safe
- Widely available and cost-effective
Sterile Water for Injection
Sterile water without a preservative is appropriate for single-use applications. It offers no antimicrobial protection, so reconstituted solutions should be used within 24 hours.
Acetic Acid (0.1%–1% Solution)
Some peptides particularly growth hormone-releasing hormones and certain GHRPs have hydrophobic regions that make them difficult to dissolve in plain water. For these, a dilute acetic acid solution (typically 0.1%–0.5%) is recommended as a primary reconstitution solvent, followed by dilution with bacteriostatic water.
Common examples where acetic acid is used:
- IGF-1 LR3
- Some GHRH analogs
- Certain collagen peptides
For researchers working with compounds like IGF-1 LR3, choosing the right initial solvent is especially important to maintain molecular integrity.

Equipment You’ll Need Before Starting
Before you begin reconstitution, gather all necessary supplies. Doing this in advance reduces the risk of contamination and procedural errors.
| Item | Purpose | Notes |
|---|---|---|
| Lyophilized peptide vial | Contains the research compound | Handle gently |
| Bacteriostatic water vial | Reconstitution solvent | Verify sterility |
| Sterile syringe (1–10 mL) | Drawing and injecting solvent | Use a fresh syringe |
| 18–21 gauge needle | Mixing/drawing solvent | Larger bore for viscous solutions |
| Alcohol prep pads (70% isopropyl) | Sanitizing rubber stoppers | Air dry before use |
| Clean workspace or sterile mat | Prevent environmental contamination | Wipe with IPA |
| Permanent marker or label | Date and identify vials | For storage tracking |
| Refrigerator (36–46°F / 2–8°C) | Storage of reconstituted solution | Never freeze |
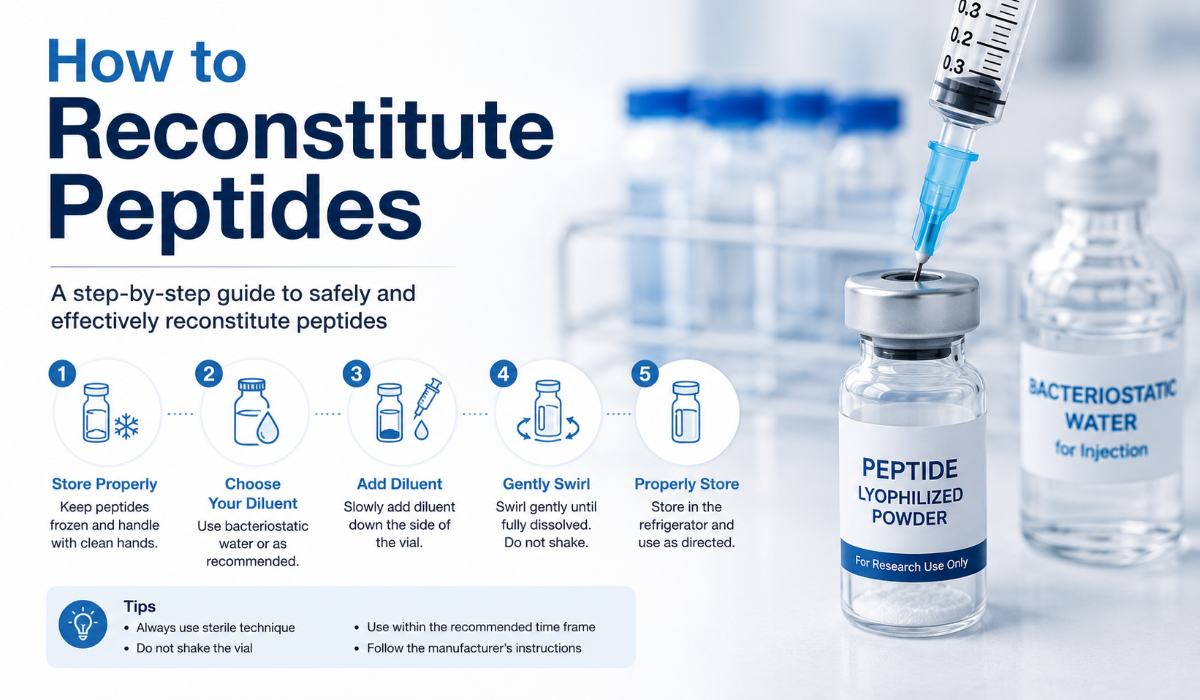
Step-by-Step: How to Reconstitute Peptides Correctly
Follow this procedure carefully. Each step serves a specific function in ensuring the safety, sterility, and integrity of your research compound.
Step 1 — Prepare Your Workspace
Wash your hands thoroughly with soap and water for at least 20 seconds. If available, use nitrile gloves throughout the process. Clean your workspace with a 70% isopropyl alcohol wipe and allow it to air dry completely. Lay out all equipment within reach before opening any vials.
Why it matters: Environmental contamination is the leading cause of compromised research solutions. Bacteria, dust, and skin oils can all degrade sensitive peptide chains.
Step 2 — Inspect the Peptide Vial
Before opening anything, inspect the peptide vial. The lyophilized powder should appear as a white or off-white cake or powder. There should be no discoloration, unusual odor, or visible contamination. Check the label for compound name, lot number, and expiration date. If the vial shows signs of damage, moisture intrusion, or unexpected color, do not use it.
Step 3 — Calculate Your Desired Concentration
This is arguably the most intellectually important step. Determining how much bacteriostatic water to add depends on the concentration you want to achieve.
The formula is simple:
Concentration (mg/mL) = Amount of Peptide (mg) ÷ Volume of Solvent Added (mL)
Example calculations:
| Peptide Amount | Water Added | Resulting Concentration | Each 0.1 mL Draws |
|---|---|---|---|
| 5 mg | 2.5 mL | 2 mg/mL | 0.2 mg |
| 5 mg | 5 mL | 1 mg/mL | 0.1 mg |
| 10 mg | 5 mL | 2 mg/mL | 0.2 mg |
| 10 mg | 10 mL | 1 mg/mL | 0.1 mg |
For a BPC-157/TB-500 10mg blend, adding 5 mL of bacteriostatic water gives a clean 2 mg/mL concentration (1 mg per component per mL), making dosage calculations straightforward in research settings.
Step 4 — Disinfect the Rubber Stoppers
Remove the plastic caps from both vials. Using a fresh alcohol prep pad, swab the rubber stopper of the peptide vial in a circular motion. Repeat with the bacteriostatic water vial. Allow both stoppers to air dry for 15–30 seconds before proceeding.
Do not blow on the stoppers to dry them. Breath contains bacteria and moisture that can contaminate your compounds.
Step 5 — Draw the Bacteriostatic Water
Attach a fresh needle to your sterile syringe. Insert the needle into the bacteriostatic water vial through the center of the rubber stopper at a 90-degree angle. Draw back the plunger to pull the calculated volume of water into the syringe.
If there is resistance (a vacuum in the vial), you can introduce a small amount of air first to equalize pressure before drawing. Remove the needle from the bacteriostatic water vial.
Step 6 — Add Water to the Peptide Vial (The Critical Technique)
This is the step where most errors occur, and it’s the one that can most directly damage your research compound.
Insert the needle into the peptide vial at a 45-degree angle. Guide the needle tip so it points toward the interior glass wall of the vial not directly onto the powder.
Inject the bacteriostatic water slowly along the side of the vial, not directly onto the powder.
This technique:
- Prevents foaming
- Avoids mechanical disruption of peptide chains
- Allows the powder to absorb the liquid without shear stress gradually
For a visual understanding of why this matters: imagine the peptide powder is a delicate sponge. Blasting water directly into it collapses the structure. Letting water flow gently down the wall allows it to wick into the powder naturally.
Step 7 — Dissolve the Peptide
After adding all the water, gently roll the vial between your palms or swirl it in a slow circular motion. Do not shake, vortex, or tap aggressively.
Most lyophilized peptides dissolve fully within 30–90 seconds of gentle swirling. The solution should appear clear to slightly opalescent. A completely opaque or particulate-laden solution may indicate:
- Incomplete dissolution (continue swirling)
- Use of the wrong solvent
- Peptide degradation
- Contamination
If the solution doesn’t clear after several minutes of gentle swirling, allow it to sit in the refrigerator for 5–10 minutes, then try again.
Never shake a peptide vial. Mechanical stress from shaking can break amide bonds that hold amino acid chains together, rendering the compound structurally incomplete for research purposes.
Step 8 — Label and Store the Reconstituted Peptide
Use a permanent marker to write on the vial:
- Date of reconstitution
- Compound name
- Concentration (mg/mL)
- Volume added
Store the labeled vial in a refrigerator set to 36–46°F (2–8°C). Most reconstituted peptides are stable for 28 days at refrigerator temperature with bacteriostatic water; some, like BPC-157 and TB-500, may retain integrity for up to 90 days under ideal conditions.
Never freeze a reconstituted peptide solution. Ice crystal formation physically shears peptide chains, causing irreversible structural damage.
Reconstitution Ratios: Quick Reference Table
Vial Size Bacteriostatic Water Concentration Achieved
| Vial Size | Bacteriostatic Water | Concentration Achieved |
|---|---|---|
| 2 mg | 1 mL | 2 mg/mL |
| 2 mg | 2 mL | 1 mg/mL |
| 5 mg | 2.5 mL | 2 mg/mL |
| 5 mg | 5 mL | 1 mg/mL |
| 10 mg | 5 mL | 2 mg/mL |
| 10 mg | 10 mL | 1 mg/mL |
| 12 mg | 6 mL | 2 mg/mL |
These are standard starting ratios used in research settings. The appropriate concentration depends on the specific compound, the protocol requirements, and the available measurement precision.
Peptide-Specific Reconstitution Notes
Different research compounds have unique characteristics that can affect how they should be reconstituted. Here’s a breakdown by category:
Growth Hormone-Releasing Peptides (GHRPs & GHRHs)
Compounds like CJC-1295/Ipamorelin generally reconstitute well in bacteriostatic water. These peptides are relatively water-soluble and dissolve readily with minimal agitation.
For researchers reviewing the complete CJC-1295/Ipamorelin research guide, proper reconstitution is a foundational prerequisite before any data collection begins.
Healing and Repair Peptides (BPC-157, TB-500)
BPC-157 (Body Protection Compound 157) and TB-500 (Thymosin Beta-4 Fragment) are among the most studied healing peptides in preclinical research. Both reconstitute readily in bacteriostatic water.
For combination research using a BPC-157 and TB-500 blend, a single reconstitution step simultaneously covers both peptides, significantly simplifying the research protocol.
When working with these compounds individually, researchers can explore the 5mg BPC-157 vial and the 5mg TB-500 vial for controlled single-compound studies.
Metabolic Research Peptides (Tesamorelin, Retatrutide, Tirzepatide)
Some metabolic and lipolytic peptides require slightly different handling. Tesamorelin, for instance, contains hydrophobic components that may benefit from acetic acid as a primary solvent before dilution. Researchers comparing tesamorelin vs sermorelin should note that both are GHRH analogs, but may differ slightly in reconstitution behavior.
For those studying newer investigational compounds, the tesamorelin 10mg visceral fat research guide provides further context.
Newer investigational triple-receptor agonists, such as retatrutide which targets the GLP-1, GIP, and glucagon pathways are typically supplied as lyophilized powders and follow standard bacteriostatic water reconstitution protocols. Researchers should always consult the compound’s Certificate of Analysis (CoA) for any special handling notes.
Similarly, those working on tirzepatide 10mg research should handle reconstitution under strict sterile conditions, as these are sensitive dual-agonist compounds.
Nootropic and Cognitive Peptides (Semax)
Semax peptide is a synthetic analog of ACTH fragments primarily studied for its neuroprotective properties. It reconstitutes easily in bacteriostatic water and is notably stable in solution. Researchers interested in its full profile should review the Semax peptide benefits guide and the comprehensive science-backed Semax benefits article.
The Semax 10mg vial is compatible with standard bacteriostatic water reconstitution at a 1–2 mg/mL concentration.
Common Reconstitution Mistakes and How to Avoid Them
Understanding what not to do is just as important as knowing the correct technique.
Mistake 1: Shaking the Vial
As emphasized throughout this guide, shaking is the single most damaging thing you can do to a reconstituted peptide. The turbulence causes mechanical degradation of peptide bonds. Always swirl gently.
Mistake 2: Injecting Water Directly onto the Powder
This causes foaming, which introduces air bubbles that can degrade the peptide through oxidation. Always direct water flow down the wall of the vessel.
Mistake 3: Using Tap Water or Saline
Tap water contains minerals, chlorine, and microorganisms. Standard saline (0.9% NaCl) is osmotically compatible but lacks the bacteriostatic properties needed for multi-dose use. Always use pharmaceutical-grade bacteriostatic water or sterile water for injection.
Mistake 4: Freezing the Reconstituted Solution
Many researchers mistakenly freeze reconstituted peptides to extend shelf life. Freezing causes ice crystal formation that physically destroys the peptide’s three-dimensional structure. Lyophilized powder can be frozen; a reconstituted solution cannot.
Mistake 5: Forgetting to Label Vials
This seems minor, but has significant implications for research validity. An unlabeled vial can lead to confusion over compound identity, concentration, or reconstitution date. Always label immediately after reconstitution.
Mistake 6: Using a Dull or Contaminated Needle
Reusing needles causes particulate contamination and increases infection risk in research settings. Always use a fresh, sterile needle for each reconstitution or draw.
Peptide Storage: Before and After Reconstitution
Pre-Reconstitution (Lyophilized Powder)
| Condition | Recommendation |
|---|---|
| Short-term (1–3 months) | Refrigerator, 36–46°F |
| Long-term (3–24 months) | Freezer, −4°F to below −20°C |
| Light exposure | Avoid direct sunlight; amber vials preferred |
| Humidity | Keep dry; store with desiccant if possible |
Post-Reconstitution (In Solution)
| Condition | Recommendation |
|---|---|
| Temperature | Refrigerator, 36–46°F only |
| Duration (bacteriostatic water) | Up to 28–90 days depending on compound |
| Duration (sterile water) | 24 hours maximum |
| Freeze? | Never |
| Light exposure | Keep in original vial; limit light exposure |
Understanding these distinctions helps researchers plan their timelines and ensures compounds remain usable throughout the full study period. Researchers working with natural peptides or peptide serums should apply the same storage principles.
How Reconstitution Affects Research Outcomes
Peptide research is only as reliable as the preparation that precedes it. A poorly reconstituted compound introduces variables that can contaminate data, reduce observed activity, or cause aggregate formation that clogs measurement equipment.
Here’s how reconstitution quality impacts specific research areas:
Muscle Growth and Recovery Research
Researchers studying the best peptides for muscle growth such as IGF-1 LR3, CJC-1295, and GHRP-6 depend on solution integrity for accurate dose-response measurements. A degraded compound produces blunted signaling responses that may be misinterpreted as low efficacy rather than poor preparation.
Fat Loss Research
Compounds studied for adipose reduction, including the best peptides for fat loss, rely on precise dosing. Inaccurate concentrations due to improper water volumes lead to under- or overdosing errors in research protocols.
Healing and Regenerative Research
The BPC-157 and TB-500 combination is widely used in tissue repair research. Maintaining the structural integrity of both peptides during reconstitution ensures that the observed angiogenic and cytoprotective effects in research models reflect the compound’s actual activity, rather than artifacts of degradation.
Facts & Research Highlights: Peptide Reconstitution
| Fact | Detail |
|---|---|
| Benzyl alcohol concentration | 0.9% in bacteriostatic water; FDA-accepted preservative concentration |
| Peptide bond vulnerability | Amide bonds can hydrolyze in extreme pH or high temperatures |
| Optimal storage temperature | 2–8°C (36–46°F) for reconstituted solutions |
| Shelf life (bacteriostatic water) | Generally 28–90 days; compound-specific |
| Lyophilized powder longevity | Up to 24 months when stored correctly at −20°C |
| Primary cause of degradation | Mechanical shearing, temperature abuse, oxidation |
| Most versatile reconstitution solvent | Bacteriostatic water (0.9% benzyl alcohol) |
| Recommended needle gauge for mixing | 18–21 gauge (smaller gauge for drawing; larger for mixing thick solutions) |
Advanced Reconstitution: Dilution and Serial Concentrations
Some research protocols require multiple concentration points for dose-response curve analysis. This requires serial dilution after the initial reconstitution.
How Serial Dilution Works
- Reconstitute the peptide at a high concentration (e.g., 2 mg/mL)
- Take an aliquot and dilute 1:1 with bacteriostatic water → 1 mg/mL
- Take an aliquot from the 1 mg/mL solution and dilute 1:1 → 0.5 mg/mL
- Continue as needed for the research protocol
Important: Each dilution step must use sterile equipment and fresh bacteriostatic water. Contamination at any step propagates through all subsequent concentrations.
For researchers studying compounds like IGF-1 LR3, serial dilution is commonly used to assess proliferative dose-response curves in cell culture research.
Reconstitution for Combination Peptide Blends
Combination vials such as the CJC-1295/Ipamorelin 10mg blend and BPC-157/TB-500 10mg blend are pre-measured blends of two research peptides in a single lyophilized vial. They are reconstituted using the same technique as single-compound vials.
Benefits of combination vials for researchers:
- Simplified reconstitution workflow (one vial instead of two)
- Pre-calculated stoichiometric ratios
- Reduced contamination risk from fewer vial entries
- Cost efficiency for long-duration studies
Researchers interested in the synergistic effects of combined peptides can consult the BPC-157 and TB-500 research overview for more details on co-administration protocols used in preclinical studies.
How Long Do Reconstituted Peptides Last?
Shelf life after reconstitution is one of the most common questions in peptide research communities and the answer depends on several variables.
Factors Affecting Post-Reconstitution Shelf Life
1. Solvent used
- Bacteriostatic water: 28–90 days
- Sterile water: 24 hours maximum
2. Peptide structure
- Disulfide-bonded peptides (like some growth factors) are more sensitive to oxidation
- Linear peptides tend to be more stable in solution
3. Storage temperature consistency
- Fluctuating temperatures shorten stability significantly
- A dedicated peptide refrigerator set to a constant 36–40°F is ideal
4. Light exposure
- UV light can break aromatic amino acid residues (Phe, Tyr, Trp)
- Store vials wrapped in foil or in a dark refrigerator
5. Number of needle entries
- Each entry risks contamination
- Use the smallest bore needle possible for drawing doses to minimize stopper fragmentation
For context on how duration and side effects interact in metabolic research, researchers can also reference the overview on how long semaglutide side effects last as a comparative model for studying compound half-lives and tolerability windows.
A Note on Research Compound Safety and Regulatory Status
All peptides from Ageless Vitality Peptides are supplied as lyophilized research chemicals, intended strictly for laboratory and investigational use. They are not approved for human consumption, clinical treatment, or therapeutic use in the United States.
Researchers should be aware that:
- FDA Regulatory Status: Peptides such as BPC-157, TB-500, and CJC-1295 are not FDA-approved drugs. They are sold as research chemicals for in vitro and preclinical research only.
- Current Research Landscape: Investigational compounds, including retatrutide and tirzepatide, are undergoing active clinical trials as of 2026. The FDA monitors their off-label or non-clinical research supply.
- Proper Sourcing: Using pharmaceutical-grade bacteriostatic water and sterile equipment is essential not just for research integrity but for maintaining proper handling standards in any lab environment.
Researchers seeking a broader overview of the peptide landscape can review the best peptides guide for a categorized summary of currently studied research compounds.
Summary: Reconstitution Best Practices at a Glance
| Step | Best Practice |
|---|---|
| Workspace prep | Clean surface, gloves, alcohol wipes |
| Solvent choice | Bacteriostatic water for multi-dose use |
| Water injection technique | Down the vial wall, not onto the powder |
| Mixing technique | Gentle swirl only; never shake |
| Concentration calculation | mg ÷ mL = mg/mL |
| Storage | 36–46°F refrigerator; never freeze |
| Labeling | Date, compound name, concentration |
| Shelf life | 28–90 days with bacteriostatic water |
| Serial dilution | Use fresh equipment at each step |
| Needle use | New sterile needle every time |
Frequently Asked Questions (FAQs)
What is the best solvent for reconstituting peptides?
Bacteriostatic water (0.9% benzyl alcohol) is the preferred reconstitution solvent for most research peptides. It provides antimicrobial protection, extends the shelf life of reconstituted solutions to 28–90 days, and is compatible with the pH range of most amino acid-based compounds. Some peptides with hydrophobic domains may require initial dissolution in a dilute acetic acid solution before being diluted with bacteriostatic water.
How much water should I add to a 5mg peptide vial?
The amount depends on your target concentration. Adding 2.5 mL of bacteriostatic water to a 5mg vial gives a concentration of 2 mg/mL. Adding 5 mL gives 1 mg/mL. For precision drawing with an insulin syringe, 1 mg/mL is often preferred because each 0.1 mL (10 units on an insulin syringe) equals 0.1 mg.
Why can’t I shake a peptide vial after adding water?
Shaking creates mechanical shear forces that can break the peptide’s amide bonds the chemical linkages between amino acids. Once these bonds are disrupted, the peptide’s three-dimensional structure is altered, and its research validity is compromised. Always swirl gently instead.
Can I freeze a reconstituted peptide?
No, Freezing a reconstituted peptide causes ice crystal formation that physically severs peptide chains. Lyophilized (dry) peptide powder can be safely stored at −20°C, but once reconstituted, it must remain refrigerated at 36–46°F and never frozen.
How long does a reconstituted peptide last in the refrigerator?
When reconstituted with bacteriostatic water, most research peptides are stable for 28–90 days at 36–46°F. Peptides reconstituted with plain sterile water have a much shorter window typically 24 hours. Always label your vials with the reconstitution date and discard if the solution becomes cloudy or shows particulates.
What needle size should I use for peptide reconstitution?
For drawing bacteriostatic water and injecting it into the peptide vial, an 18–21 gauge needle is appropriate. The larger bore allows smooth fluid transfer. For subsequent draws from the reconstituted vial (if used in research protocols), a finer needle reduces particulate contamination at the vial stopper.










